I've been baking for just under a year using the starter from Tartine Bread. I've always used water from my Brita pitcher and have had no problems until recently. One day I noticed that the bottom of my pitcher was a little green, looking up online a lot of people seem to be having problems with algae growing in their brita pitchers. The day prior to realizing this, I fed my starter using this water and ever since that day my starter looks like this about a day after I feed it:

A closeup reveals weird looking strands.
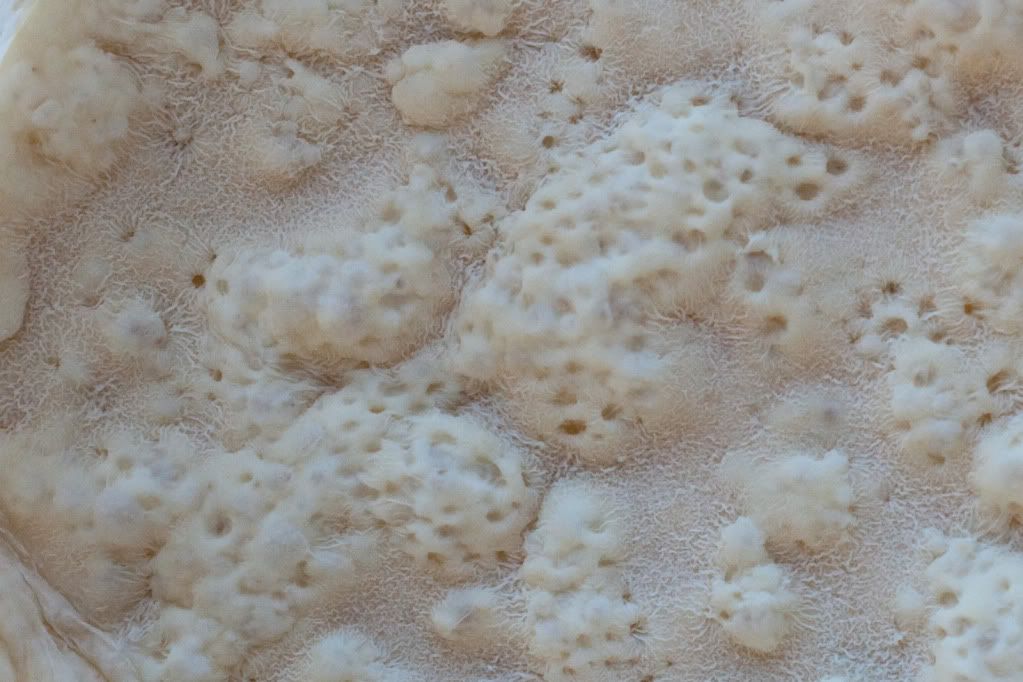
I have used the starter a few times since this has happened and it works just fine, it rises and falls after a feeding, smells normal but it just looks like...well this. I'm just worried that I should toss this batch and begin a new starter. I have thoroughly cleaned the pitcher but I wish I would have noticed the green substance before feeding my starter. Has anyone else run into this sort of situation? Would feeding it pineapple juice resolve the problem? Any help would be greatly appreciated. Thank you!
First off, I would start a new starter using Debra Wink's pineapple juice solution immediately. This is so that, if you try to save THIS starter, and it fails, your new starter will already be several days under way. Using Debra's complete instructions (from pineapple juice part 2), you'll be good for about a week or so.
After that's on the counter and starting, you might try to save your old starter by 'washing' it, which simply means, use an -extremely- small sample of the old starter, taken from like the middle of the polluted culture, and adding it to a normal feed. This small sample can be 1 gram or even less, to say, 40 or 60 grams of refreshment. Use a different container (obviously), and I'd recommend plain old tap water at first. Something slightly chlorinated and hardened with minerals will probably help combat whatever cheesiness or mold may be in the small sample. Watch your refreshed starter and refresh again with a normal sample (say 2:1:1) after it's at least doubled, but not peaked. Do this for a day or two, and then revert back to 1:1:1 maintenance every 12 hours (let it peak and crash) and see if behaves and smells correctly. You can also go back to a filtered or bottled water. This old starter, if able to be saved, should be back to normal health within 3-5 days. If at any point you still see signs of pollution, I'd pitch it and focus on the new starter. If you save it, you can go back to your usual maintenance and storage procedures you're comfortable with.
And Hi Dan! Good to see you... = ]
- Keith
Have you eaten bread baked with the starter after it started looking like this? From your statements it seems like you have, so on to question two...
Did you get sick from it?
The vast majority of food poisoning is instant or very nearly so. You should feel an effect of some kind within 24 hours. On the outside, certain rare kinds of foodborne illness can produce symptoms after 3 days or more. These illnesses are from meat and pathogens borne by livestock or, much more often, wild game.
If it's not making you sick, I wouldn't worry about it. But that's me.
EDIT: If you're nervous, toss it. You can always make a new starter :)
If your interest is in keeping that year old starter going because it is a year old.....lets face it, it's fun to say, my starter is xxx number of years old, then why don't you just save a little bit, like 1/4 cup or even a few tablespoons and start fresh feeding it..........you can start another new batch just in case.......all you waste is a little time and a little flour...
My thoughts are that your starter is perfectly safe and even if it had a little alge it it, it would probably not be harmful. I'm no authority and I'm just guessing.
If your interest is in keeping that year old starter going because it is a year old.....lets face it, it's fun to say, my starter is xxx number of years old, then why don't you just save a little bit, like 1/4 cup or even a few tablespoons and start fresh feeding it..........you can start another new batch just in case.......all you waste is a little time and a little flour...
My thoughts are that your starter is perfectly safe and even if it had a little alge it it, it would probably not be harmful. I'm no authority and I'm just guessing.
Thank you all very much for your advice and comments. I have just decided to begin a new starter, it's going to take a while so for the time being I'm just going to continue using this starter until the new one is ready (we eat a lot of bread in my house, about 5-6 boules a week).
I didn't know that I could sterilize using vinegar (which I will do from now on) so I sterilized the pitcher by soaking it in a bleach solution, I didn't want to take any chances. My previous pitcher (which was a few years old) had the same fate so I just recently bought this pitcher; I don't know what is causing this.
Mr. Robertson's method of maintaining the starter is pretty much "washing" it everytime bread is baked. His method states to take a tablespoon of old starter and adding it to a large portion of flour and water, only half of which is used to bake the bread, the other half is the new starter. I've done this many times and everytime my starter still becomes this hideous and veiny thing.
I haven't gotten sick from consuming it and it tastes just fine. Thanks again folks!
How much is "a large portion of flour and water" that you are adding this tablespoon of starter to?
it's 1 TBSP of starter to 200 grams flour (50/50 A.P and whole wheat) and 200 grams water.
JimmyChoCho,
I hope you're still monitoring this thread. Back when your post appeared, I had my suspicions about what it was, but I was unable to find any reference photos to support them. Last night, I once again scoured the Internet for images of this phenomenon for a project I'm currently working on. What I'm talking about is the pseudohyphal form of yeast. Saccharomyces and Candida---the yeasts most commonly found in sourdoughs---are dimorphic, meaning that they have two distinctly different modes of growth. In bread-making they generally grow as single-cell yeast that we’re most familiar with. But when nutrients are depleted—nitrogen in particular—they may switch to the filamentous, pseudo-hyphal form. (True hyphae are the hairlike filaments of molds.) And guess what---algae consume nitrogen. So I suspect the yeast may have reverted to this "pseudo mold" form because the algae robbed its nitrogen sources. While it looks very strange, it is still the same harmless microorganism. I like that you describe it as "veiny", because that's exactly the term that came to mind when I looked at images such as these:
http://howardhughes.trinity.duke.edu/uploads/assets/image/Christine Kim/colony morph
Above shows a close-up of one small colony each of S. cerevisiae---yeast form (left) and pseudohyphal form (right). Various Candida species I found look the same, such as this C. albicans:
http://openi.nlm.nih.gov/imgs/rescaled512/2739428_pgen.1000664.g004.png
Zooming out a bit more:
http://classconnection.s3.amazonaws.com/237/flashcards/725237/jpg/b2500194-food_spoil
This one is Candida krusei. The colonies have spread and grown into each other more, making it easier to see the similarity in texture to your photo above.
The areas that look like corals rising up throughout in your photo are probably just where the CO2 gases are bubbling up underneath this blanket, no?
Herein lies a problem. The Robertson method of diluting a starter which probably is never allowed to build up any significant acidity to begin with, may be raising the pH into the range that allows growth of this algae. Especially if your tap water is alkaline as well, as many are. This may be a rare combination of factors, but the result is kinda cool to see. Thank you for posting these photos.
I know you started over, but if you find it happening again, it shouldn't be too difficult to banish algae from a starter by using algae-free water, letting the acids accumulate so that the pH becomes inhibiting, and keep the starter in the dark until things turn around for you.
How have things been going since?
dw
Hi Debra,
Not Jimmy here, but thanks for sharing your research again, fascinating! I think I may have seen this on a starter of mine in the past, either in a creation state, or a too-long-in-the-fridge state. Nice to know that it's not harmful.
Debra,
This is great information, thanks for following up.
Peter
Hi! I just posted about an issue i just discovered with my starter that had been left in the fridge for about a month. I'm putting all the pieces together and I think this thread has solved my problem. It sounds you have a good grasp on what this growth is and I wondered if you could help me pin it down. I too used the Robertson Tartine starter method and have baked with it twice and then refrigerated it for a month. The lid was on much tighter than i thought when I returned to it and I was surprised to find these strange formations. The smell is mild and yeasty at most. I immediately thought it was harmless just based on the coloration but I was too afraid to just scrape it off and revive the starter. Could you take a look at this picture and perhaps weigh in on how you would proceed? Also the algae bit threw me off because I used still bottled mineral water so I thought that would be a stretch. I thought your theory on the gases layering was perhaps right on since the lid was on tighter than usual. Thanks I appreciate any thoughts!
Debra,
Thanks for recalling this topic. Very interesting indeed and the slides you posted are beautiful....amazing stuff this is that we couldn't bake our loaves without and how ingenious of it to change forms when it's food source changes.....Once again me thinks/knows we humans certainly are not the most likely to survive on this planet in the long run....My 16 year old son refuses to eat anything different :-O He'd be first to go and I would follow shortly after *-)
Thanks for taking the time to explain this in a way that someone like me can grasp it too. Will take my walk now and marvel at the wonders of the microscopic world that keeps us all alive.
Take Care,
Janet
It will be the coolest bread book ever. Awesome read; thanks. Never knew the yeast we encounter in baking was polymorphic, or that algae may trigger the polymorphism.
I remember seeing it weeks into refrigeration with an untouched tightly covered 100% hydration rye starter. I would have thought the slowly increasing acid would keep the starter "in check."
Mini, keep in mind that this yeast trait isn't directly influenced by pH. Most sources site insufficient nitrogen as the primary cause, especially when there are plenty of sugars available.
...I'm thinking that as CO2 gas is coming off the starter, it is the heavier gas and any loose nitrogen is forced to the top of the container just under the lid.
Yeasts get their nitrogen from ammonium ions and free amino acids. They don't break down proteins, so proteolytic enzyme activity in flour would be something to look at in the absence of other conspicuous causes.
Best wishes,
dw
Debra, I saw this recent reply of yours on this old thread. This is exactly what I found on my starter the other day, the morning after baking using that same starter. It alarmed the daylights out of me. So you're saying this is a secondary formation of the naturally occurring yeasts in a starter when nitro is depleted?
I left the lid on my jar "ajar" (no pun intended).... after my bake, without feeding it until later on the following day. I keep my house warm.
I didn't not view this as mold, based on your info and I simply left it in my jar, and fed the starter.
As long as this isn't true mold, it'll be okay? I'm soooo glad you posted this wild info.
I posted that in 2013, but the time stamp changed when I went in to edit the links. Unfortunately, they no longer take you to the original photos, so I'm so glad I captured them at the time. Things like that can be elusive :)
I wouldn't worry about seeing that on your starter unless it persists. If it appears frequently, even with regular and adequate feeding then there's a nutritional deficiency that needs investigating. Before your bake, had this starter been in the refrigerator a while?
Hi,
I encountered the same problem. I searched the internet and found your answer. I realized that in the last time i used some special alcaline water with a 9.2 PH. This could just confirm your theory. The picture i added is just 1-2 days after the mold formed. If i wait 1-2 more days it becomes exactly as above.
I will try to fight it by using another water.
I've never heard of that before. I use a Brita-type filtre bottle and have never seen anything resembling algae in it.